A brand of innovative IVD products established by PBF Diagnostics
Focus on product development of infectionand oncology diseases
Market ISO13485, CE mark and TFDAcertified rapid tests
Services for test development andcontract manufacturing provided
Based in Taiwan, and think globally
|
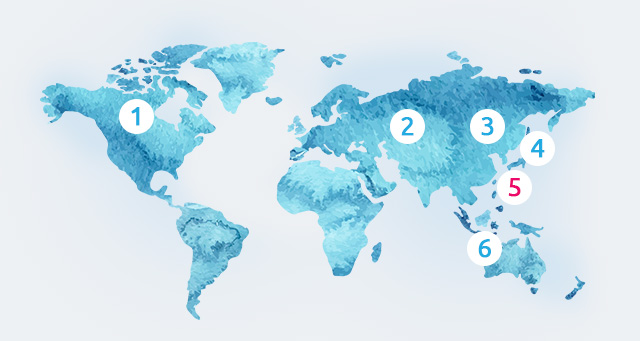
US |
|
Europe |
|
CHINA |
|
Japan |
|
TAIWAN |
|
Asia Pacific |
PBF Diagnostics is able to deliver reliable products
to meet all customers’ demands including OEM/ODM projects
No.308-8, Sec. 1, Datong Rd., Xizhi Dist., New Taipei City 22146 Taiwan
Tel: +886-2-2691-9895
Fax: +886-2-2691-9575
Email: [email protected]
16F., No.3, Park St., Nangang Dist., Taipei City 115, Taiwan
www.pbf.com.tw